04 Mar 10:36
by Surendran Amrutha, Sankaran Radhika and Gopinathan Anilkumar
Abstract
Iron- and cobalt-catalyzed Sonogashira coupling reactions are becoming central areas of research in organic synthesis. Owing to their significant importance in the formation of carbon–carbon bonds, numerous green and nanoparticle protocols have emerged during the past decades. The non-toxic and inexpensive nature of catalysts gained much attention in recent times. In this context, their catalytic nature and activity in Sonogashira coupling reactions were well explored and compared. Most importantly, one of the highlights of this review is the emphasis given to green strategies. This is the first review on iron- and cobalt-catalyzed Sonogashira coupling reactions which comprehends literature up to 2020.
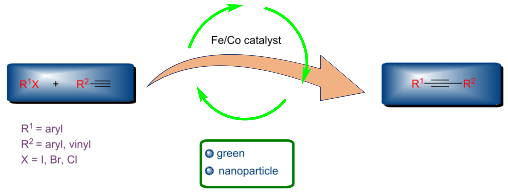
Beilstein J. Org. Chem. 2022, 18, 262–285. doi:10.3762/bjoc.18.31
01 Mar 16:49
by Masahiro Abe, Sayasa Nitta, Erina Miura, Tetsutaro Kimachi, and Kiyofumi Inamoto

The Journal of Organic Chemistry
DOI: 10.1021/acs.joc.1c03011
08 Dec 13:11
by Jin Zhang,
Pei Zhang,
Lei Shao,
Ruihong Wang,
Yangmin Ma,
Michal Szostak

The first mechanochemical strategy for highly chemoselective, solvent-free palladium-catalyzed cross-coupling of amides by N−C bond activation is reported. The protocol advances the biorelevant amide bond cross-coupling manifold to solid-state solventless cross-coupling methods.
Abstract
Although cross-coupling reactions of amides by selective N−C cleavage are one of the most powerful and burgeoning areas in organic synthesis due to the ubiquity of amide bonds, the development of mechanochemical, solid-state methods remains a major challenge. Herein, we report the first mechanochemical strategy for highly chemoselective, solvent-free palladium-catalyzed cross-coupling of amides by N−C bond activation. The method is conducted in the absence of external heating, for short reaction time and shows excellent chemoselectivity for σ N−C bond activation. The reaction shows excellent functional group tolerance and can be applied to late-stage functionalization of complex APIs and sequential orthogonal cross-couplings exploiting double solventless solid-state methods. The results extend mechanochemical reaction environments to advance the chemical repertoire of N−C bond interconversions to solid-state environmentally friendly mechanochemical methods.
07 Dec 13:23
by Haonan Xiang, Jie Liu, Jieping Wang, Lvqi Jiang, and Wenbin Yi

Organic Letters
DOI: 10.1021/acs.orglett.1c03813
04 Nov 08:40
by Michael D. Mandler, Andrew P. Degnan, Shasha Zhang, Darpandeep Aulakh, Ketleine Georges, Bhupinder Sandhu, Amy Sarjeant, Yeheng Zhu, Sarah C. Traeger, Peter T. Cheng, Bruce A. Ellsworth, and Alicia Regueiro-Ren

Organic Letters
DOI: 10.1021/acs.orglett.1c03201
01 Oct 08:40
by Murugaiah A. M. Subbaiah and Nicholas A. Meanwell

Journal of Medicinal Chemistry
DOI: 10.1021/acs.jmedchem.1c01215

30 Sep 08:26
by Zhiwei Zhao, Ge Zeng, Yinan Chen, Jinming Zheng, Zhongyan Chen, Yinlin Shao, Fangjun Zhang, Jiuxi Chen, and Renhao Li

Organic Letters
DOI: 10.1021/acs.orglett.1c02962

14 Jul 11:38
by Pezhman Shiri, Ali Mohammad Amani and Thomas Mayer-Gall
Abstract
Diverse strategies for the efficient and attractive synthesis of a wide variety of relevant 1,4,5-trisubstituted 1,2,3-triazole molecules are reported. The synthesis of this category of diverse fully functionalized 1,2,3-triazoles has become a necessary and unique research subject in modern synthetic organic key transformations in academia, pharmacy, and industry. The current review aims to cover a wide literature survey of numerous synthetic strategies. Recent reports (2017–2021) in the field of 1,4,5-trisubstituted 1,2,3-triazoles are emphasized in this current review.

Beilstein J. Org. Chem. 2021, 17, 1600–1628. doi:10.3762/bjoc.17.114
23 Mar 09:34
by Hai-Ming Wang, Chao-Dong Xiong, Xiao-Qu Chen, Chun Hu, and Dong-Yu Wang

Organic Letters
DOI: 10.1021/acs.orglett.1c00504

19 Mar 16:46
by Devaneyan Joseph, Muhammad Aliyu Idris, Jiajia Chen, and Sunwoo Lee

ACS Catalysis
DOI: 10.1021/acscatal.0c05690

18 Mar 14:44
by Hao Wang, Zeyu Xi, Shuai Huang, Rui Ding, Yang Gao, Yongguo Liu, Baoguo Sun, Hongyu Tian, and Sen Liang

The Journal of Organic Chemistry
DOI: 10.1021/acs.joc.0c02768

17 Mar 15:35
by Mingjie Wei,
Dacheng Liang,
Xiaohui Cao,
Wenjun Luo,
Guojian Ma,
Zeyuan Liu,
Le Li

1‐Hydroxybenzotriazole (HOBt) was identified as an efficient nucleophilic catalyst to activate sulfonyl fluorides and fluorosulfates for a broad‐spectrum, catalytic amidation to prepare various sulfonamides and sulfamates, including sterically hindered ones. The potential of this methodology in medicinal and process chemistry has been demonstrated by multidecagram‐scale synthesis of an amantadine derivative and synthesis of the drug Fedratinib.
Abstract
A broad‐spectrum, catalytic method has been developed for the synthesis of sulfonamides and sulfamates. With the activation by the combination of a catalytic amount of 1‐hydroxybenzotriazole (HOBt) and silicon additives, amidations of sulfonyl fluorides and fluorosulfates proceeded smoothly and excellent yields were generally obtained (87–99 %). Noticeably, this protocol is particularly efficient for sterically hindered substrates. Catalyst loading is generally low and only 0.02 mol % of catalyst is required for the multidecagram‐scale synthesis of an amantadine derivative. In addition, the potential of this method in medicinal chemistry has been demonstrated by the synthesis of the marketed drug Fedratinib via a key intermediate sulfonyl fluoride 13. Since a large number of amines are commercially available, this route provides a facile entry to access Fedratinib analogues for biological screening.
08 Mar 07:56
by Tiffany Piou, Yuriy Slutskyy, Nancy J. Kevin, Zhongxiang Sun, Dong Xiao, and Jongrock Kong

Organic Letters
DOI: 10.1021/acs.orglett.1c00100

01 Mar 14:33
by Chen, Wanli
Synlett
DOI: 10.1055/s-0037-1610766

Copper-catalyzed reactions of N-propargyl-N-(2-iodoaryl)amides with sodium azide have been developed, providing a novel and efficient method for the synthesis of [1,2,3]triazolo[1,5-a]quinoxaline compounds under mild conditions in moderate yields by tandem copper-catalyzed azide-alkyne cycloaddition and Ullmann-type coupling.
[...]
Georg Thieme Verlag KG Rüdigerstraße 14, 70469 Stuttgart, Germany
Article in Thieme eJournals:
Table of contents | Abstract | Full text
25 Feb 12:12
by Zbigniew Malinowski, Emilia Fornal, Agata Sumara, Renata Kontek, Karol Bukowski, Beata Pasternak, Dariusz Sroczyński, Joachim Kusz, Magdalena Małecka and Monika Nowak
Abstract
Amino- and polyaminophthalazinones were synthesized by the palladium‐catalyzed amination (alkyl- and arylamines, polyamines) of 4-bromophthalazinones in good yields. The coordinating properties of selected aminophthalazinones towards Cu(II) ions were investigated and the participation of the nitrogen atoms in the complexation of the metal ion was shown. A biological screening of the potential cytotoxicity of selected synthesized compounds on HT-29 and PC-3 cell lines, as well as on the L-929 cell line, proved that some amino derivatives of phthalazinone show interesting anticancer activities. The detailed synthesis, spectroscopic data, and biological assays are reported.
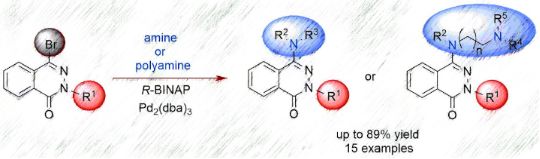
Beilstein J. Org. Chem. 2021, 17, 558–568. doi:10.3762/bjoc.17.50
25 Feb 09:52
by Olga Bakhanovich, Viktor Khutorianskyi, Vladimir Motornov and Petr Beier
Abstract
The rhodium-catalyzed transannulation of N-perfluoroalkyl-1,2,3-triazoles with aromatic and aliphatic terminal alkynes under microwave heating conditions afforded N-perfluoroalkyl-3,4-disubstituted pyrroles (major products) and N-fluoroalkyl-2,4-disubstituted pyrroles (minor products). The observed selectivities in the case of the reactions with aliphatic alkynes were high.
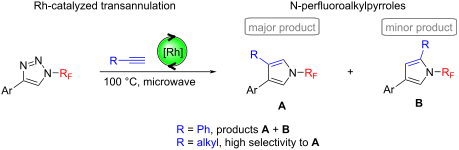
Beilstein J. Org. Chem. 2021, 17, 504–510. doi:10.3762/bjoc.17.44
No more posts. Check out what's trending.















